Two Google AI models. Names that sound similar. And a choice that, depending on the nature of your work, could have real implications on data governance.
If you work in pharma, medtech, medical affairs, or healthcare communication, you have probably seen Gemma 4 and Gemini mentioned together, often without a clear explanation of what sets them apart.
Both are Google. Both are powerful. However, they are designed for fundamentally different contexts, and this distinction becomes crucial when patient data, unpublished trial results, or commercially sensitive documents are involved.
At MedTEXTpert, we assess AI tools based on their effectiveness for healthcare and life science professionals, ensuring safety and compliance. This post gives you the practical breakdown.
The Core Difference: Where Does Your Data Go?
Gemini is Google’s cloud-based AI platform. When you use it — via the web app, Google Workspace, or the API — your prompts travel to Google’s servers, are processed there, and the response is returned to you. That is not a flaw; it is how cloud AI works. But for many tasks in healthcare, it is a critical factor to understand before you start typing.
Gemma 4, released on 2 April 2026 under an Apache 2.0 open-source license, works entirely differently. You download the model weights, run them on your own hardware — a workstation, a local server, or even a capable laptop — and nothing leaves your infrastructure. No prompts. No documents. No data of any kind is transmitted externally. This is what distinguishes local deployment from accessing – ATTENTION – Gemma 4 through cloud-based interfaces such as Google AI Studio, where standard cloud data processing applies.
When Gemini works well in healthcare and medcomms
Gemini’s strengths are scale, integration, and multi-modal capability. For work involving publicly available information — synthesising guidelines, reviewing published literature, or producing educational content from open-access data — it is a genuinely powerful and accessible tool.
1) Visual Content Creation with Nano Banana 2
One immediately useful capability for medcomms professionals is the image generation now built into Gemini. Released in February 2026, Nano Banana 2 (the technical name is Gemini 3.1 Flash Image) produces high-resolution visuals — up to 4K — with legible, accurately rendered text directly within images. For a field where scientific precision in diagrams is non-negotiable, this matters.
A practical example: a medical education team preparing congress materials could prompt Gemini to generate a mechanistic pathway diagram or a disease staging infographic with correctly labeled annotations — without involving a graphic designer in the initial draft.
Gemini can also read existing images. Photograph a hand-drawn diagram from an advisory board meeting, and it can convert it into a structured digital table or a professional slide layout.
2) Processing Large Volumes of Published Scientific Content
Gemini’s context window—up to 1 Mio tokens in the Advanced tier—makes it well suited to synthesizing large bodies of published text. (Read here if you want to learn about tokens.)
A practical example: a medical affairs professional preparing a competitive landscape summary could upload multiple clinical guidelines or congress abstracts and ask Gemini to extract and compare specific endpoints, safety signals, or dosing protocols across them.
Where the input is already in the public domain, the cloud processing model is perfectly appropriate.
3) Google Workspace Integration
For teams already working in Google Docs, Slides, or Gmail, Gemini reduces friction considerably. Drafting a summary of a published meta-analysis, structuring meeting notes, or building a slide starter set directly within familiar tools has real practical value for busy medical affairs or communications teams.
Where Gemma 4 Changes the Equation for Healthcare
Gemma 4 becomes the right answer the moment your work involves data that cannot, or should not, leave your own infrastructure. The compliance logic is straightforward: under GDPR, transferring personal data to third-country servers — including US-based cloud providers — requires formal Data Processing Agreements, Transfer Impact Assessments, and Standard Contractual Clauses. When an open-weight model runs locally, that obligation largely disappears, because no data crosses a boundary in the first place.
There is also a further legal dimension that deserves more attention than it typically receives. The US CLOUD Act grants federal authorities the power to compel US-headquartered technology companies to produce data stored on their servers, regardless of where those servers are physically located. For European healthcare organisations processing sensitive patient or trial data, this creates a compliance tension that is genuinely difficult to resolve within the existing legal architecture — and that local execution sidesteps entirely.
Possible Real-World Scenarios *
- Congress and symposium content based on unpublished data.
A medcomms writer building a satellite symposium slide deck around phase III results that have not yet been published is working with some of the most commercially sensitive material in the industry. Running Gemma 4 locally to assist with structuring arguments, drafting speaker notes, or checking narrative consistency means that data stays entirely within the agency or client infrastructure — not on a cloud server governed by a third-party provider’s terms.
- Publication planning and manuscript drafting.
When a publications team is working with unpublished study data — preparing a first author brief, drafting a methods section, or building a gap analysis against the existing literature — that content cannot go through a cloud tool without triggering data governance questions. Gemma 4 handles the drafting and iteration locally, while Gemini remains perfectly appropriate for scanning what is already in the public domain.
- Agency claims and messaging libraries.
A medcomms agency maintaining a proprietary claims matrix or approved messaging framework for a pharma client holds genuinely valuable intellectual property — theirs and their client’s. Running Gemma 4 locally to cross-reference new content against that library, flag potential off-label statements, or adapt global messaging for a local market means neither the agency’s IP nor the client’s approved content leaves the building. The model can even be fine-tuned on the specific style and scientific standards of a therapeutic area over time.
- Medical education content for HCPs.
Developing a disease awareness module, an MSL scientific exchange deck, or a KOL advisory board pre-read often involves combining published data with client-specific positioning and unpublished insights. The published component can be researched with Gemini; the drafting and refinement of the final content — which blends proprietary strategy with science — belongs in a local environment.
- Internal training and launch readiness materials.
Product launch briefing packs, therapy area onboarding modules for new MSLs, and internal FAQs for field medical teams all contain commercially sensitive pipeline information well before it reaches the public domain. Gemma 4 makes it possible to draft, iterate, and refine that content without exposing it to external infrastructure.
It is worth noting that Gemma 4’s hardware requirements are more accessible than many assume. Its Mixture-of-Experts architecture means the larger 27B model can run on a single high-specification workstation (no data center needed), making local deployment genuinely practical for agencies and in-house teams alike.
Side-by-Side: Choosing the Right Tool for the Task*
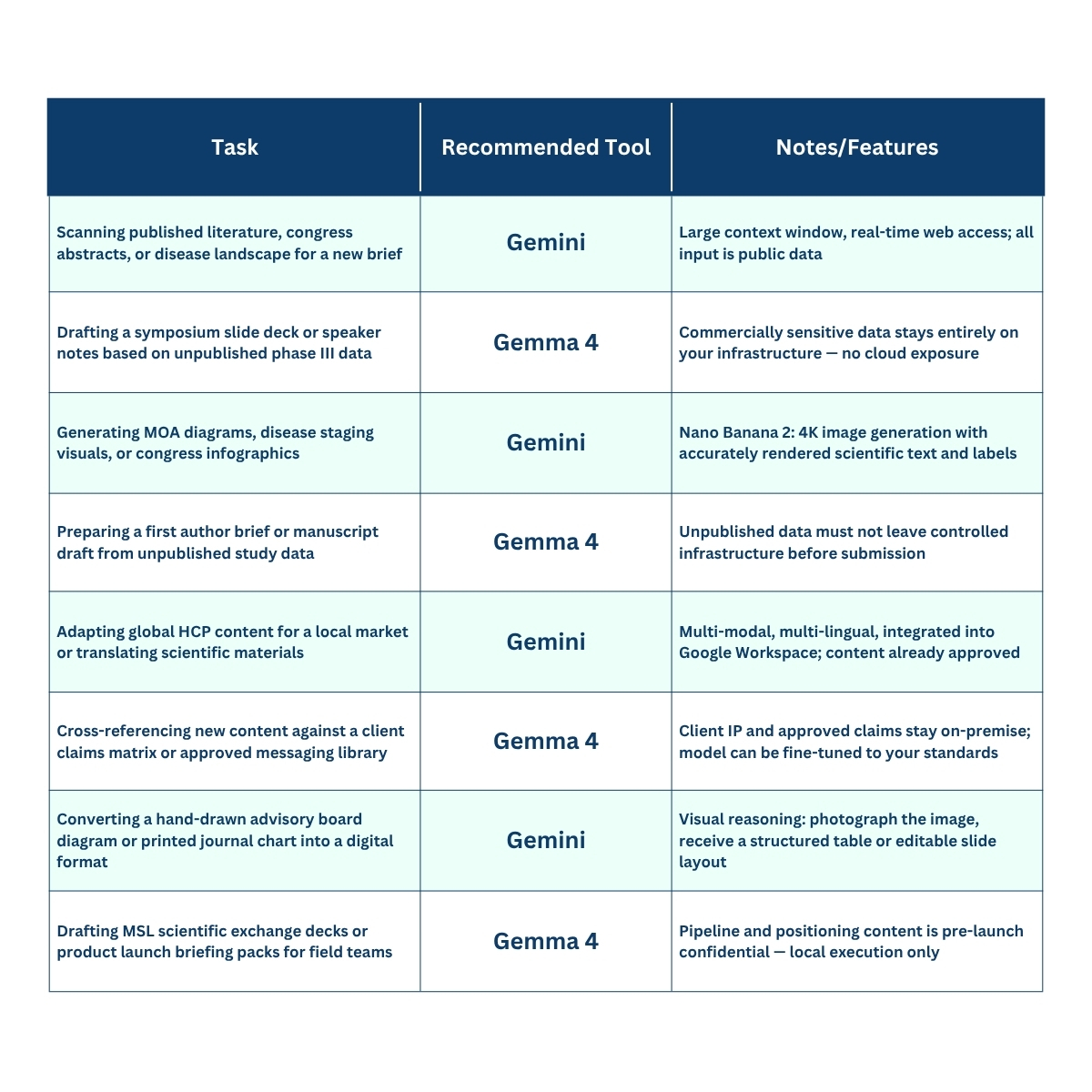
* The examples in this table reflect what Gemma 4 is designed to do — and we are aware that some of them will spark debate. Running AI locally does not automatically make a workflow compliant. The suitability of any tool for a specific task will always depend on your organisation’s data governance policies, client agreements, and applicable regulations (This is also applies to Gemini or other AI-tool.)
What we are saying is this: if Gemma 4 delivers on its promise of robust local execution, these are the use cases where that capability becomes genuinely relevant for medcomms teams. As always, human oversight, validation, and professional judgement remain non-negotiable.
A Note on Gemini’s Compliance Framework
Gemini is not inherently non-compliant — and it is important to be precise about this. Google has established a meaningful compliance framework: Gemini for Workspace processes European users’ data under GDPR via Google Ireland Ltd., includes Standard Contractual Clauses for cross-border transfers, and achieves HIPAA compliance through Workspace and Vertex AI under a signed Business Associate Agreement.
The challenge arises at the edges — when data is processed through Gemini API endpoints outside a formal Workspace or Vertex agreement, or when Transfer Impact Assessments have not been completed, or when the US CLOUD Act’s potential reach creates residual uncertainty for sensitive European healthcare data. These are not hypothetical edge cases; they are the everyday reality of most medcomms teams working across borders and therapeutic areas.
Gemma 4 removes that uncertainty by removing the transfer. For teams where the compliance officer needs a simple, auditable answer to “where is this data processed?”, local execution provides one: here, on our infrastructure, by no one else.
The Practical Answer: Use Both, Deliberately
The most effective medcomms and medical affairs teams in 2026 are not asking “which AI should we use?” They are asking “which AI should we use for this specific task?” The distinction is everything.
A practical workflow might look like this: a medical writer uses Gemini to scan published literature and pull together an initial synthesis of the public evidence base for a new therapeutic area. When it comes time to cross-reference that evidence with unpublished study data, draft sections of a regulatory submission, or query internally approved messaging against a claims database — that work moves to a local Gemma 4 instance. The cloud tool handles what is public and fast; the local tool handles what is confidential and critical.
This is not an either/or decision. It is a routing decision — and making it deliberately, with a clear understanding of what data each task involves, is what separates a compliant AI workflow from an avoidable risk.
Frequently asked questions
What is the difference between Gemma 4 and Gemini?
Gemini is a cloud-based AI platform, while Gemma 4 is an open-weight model that can run locally on your own hardware. For healthcare teams, that affects privacy, compliance and data governance.
Is Gemini compliant for healthcare use?
Gemini can be used within structured compliance frameworks, but suitability depends on the deployment model, contracts, transfer assessments and the type of data being processed.
When should healthcare teams use Gemma 4 instead of Gemini?
Gemma 4 is more relevant when work involves unpublished trial data, proprietary claims libraries, confidential launch materials or other content that should remain inside controlled infrastructure.
Not sure how to structure a compliant AI workflow for your team?
At medTEXTpert, we help pharma, medtech, and medical communications teams build practical, GDPR-aware AI workflows — from tool selection and compliance mapping to hands-on integration workshops tailored to your specific therapeutic area and content types.
We work across the full spectrum: open-weight local models, cloud platforms, and hybrid architectures. Our approach is always tool-agnostic, human-validated, and built on 25+ years of combined expertise in clinical, regulatory, and marketing communication.
Visit -> https://www.medtextpert.com/ai-in-medical-communications/ to explore our services — or reach out directly to discuss what your team needs.
AI Disclosure & Professional Statement
AI tools were used to support research and initial drafting; all content has been reviewed, edited, and validated by the author. MedTEXTpert takes full responsibility for accuracy and scientific integrity.
Note: The tools mentioned here are not intended for clinical diagnosis or medical advice.




